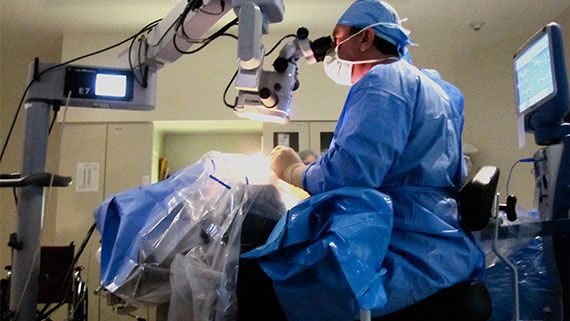
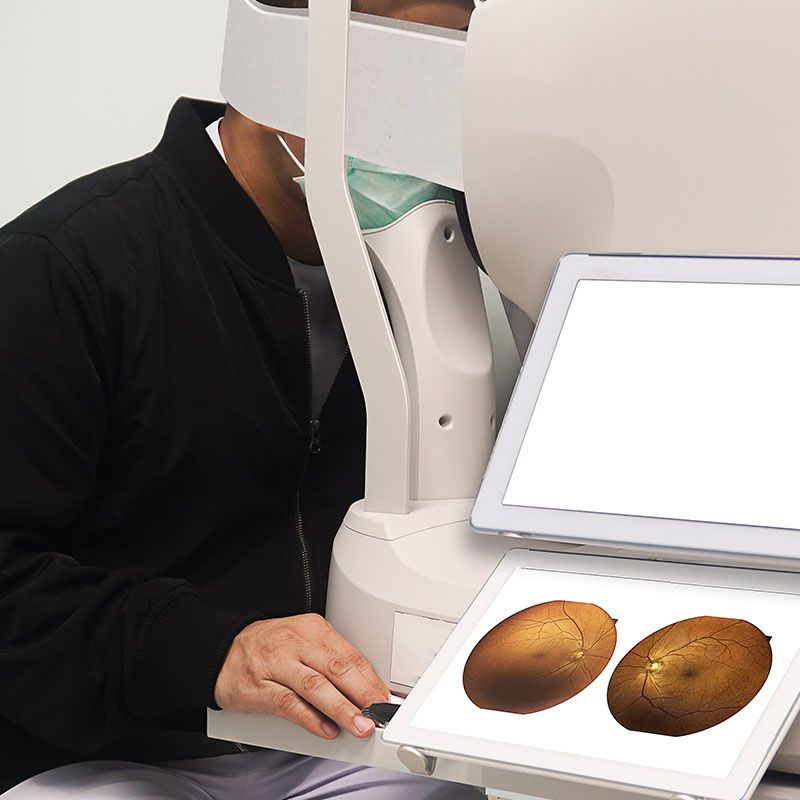
Dr. McCabe specializes in State-of-the-Art "custom cataract surgery." Employing wavefront technology, he selects the intraocular lens implant that is best for each patient's eye and specific vision needs resulting in sharper vision and greater satisfaction.

PERSONAL & PROFESSIONAL EYE CARE
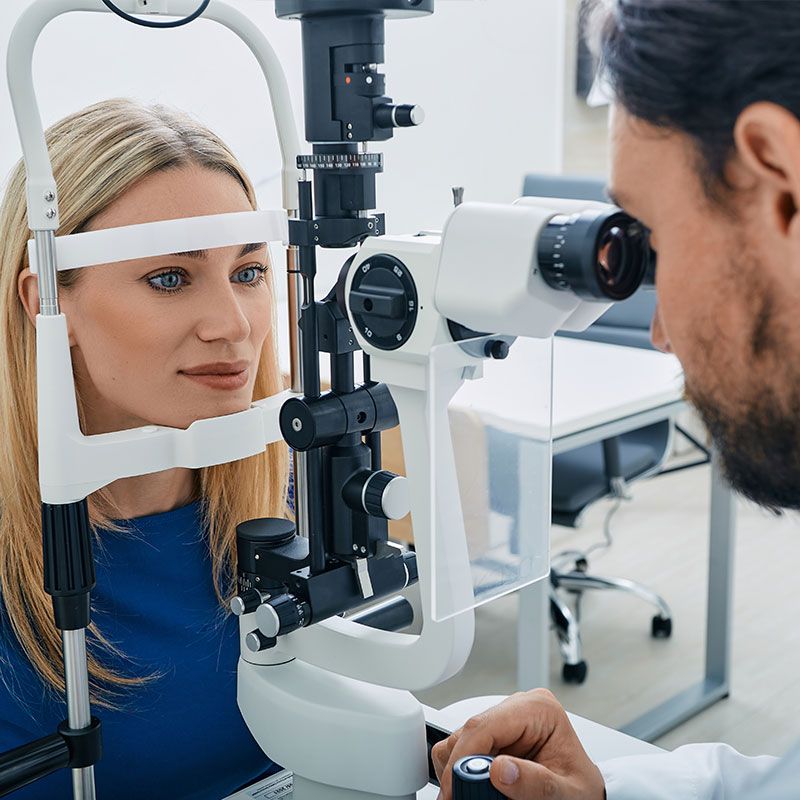
Welcome to our practice, McCabe Vision Center. Our team of experienced doctors is dedicated to helping you achieve optimal vision and eye health. We offer a wide range of services to help you maintain healthy eyes for life. We look forward to serving you and your family.
Learn Morefeatured services


Insurance We Accept










READY TO SCHEDULE AN APPOINTMENT?
Complete the form below to request an appointment. Our team will connect with you to confirm your requested appointment date and time.
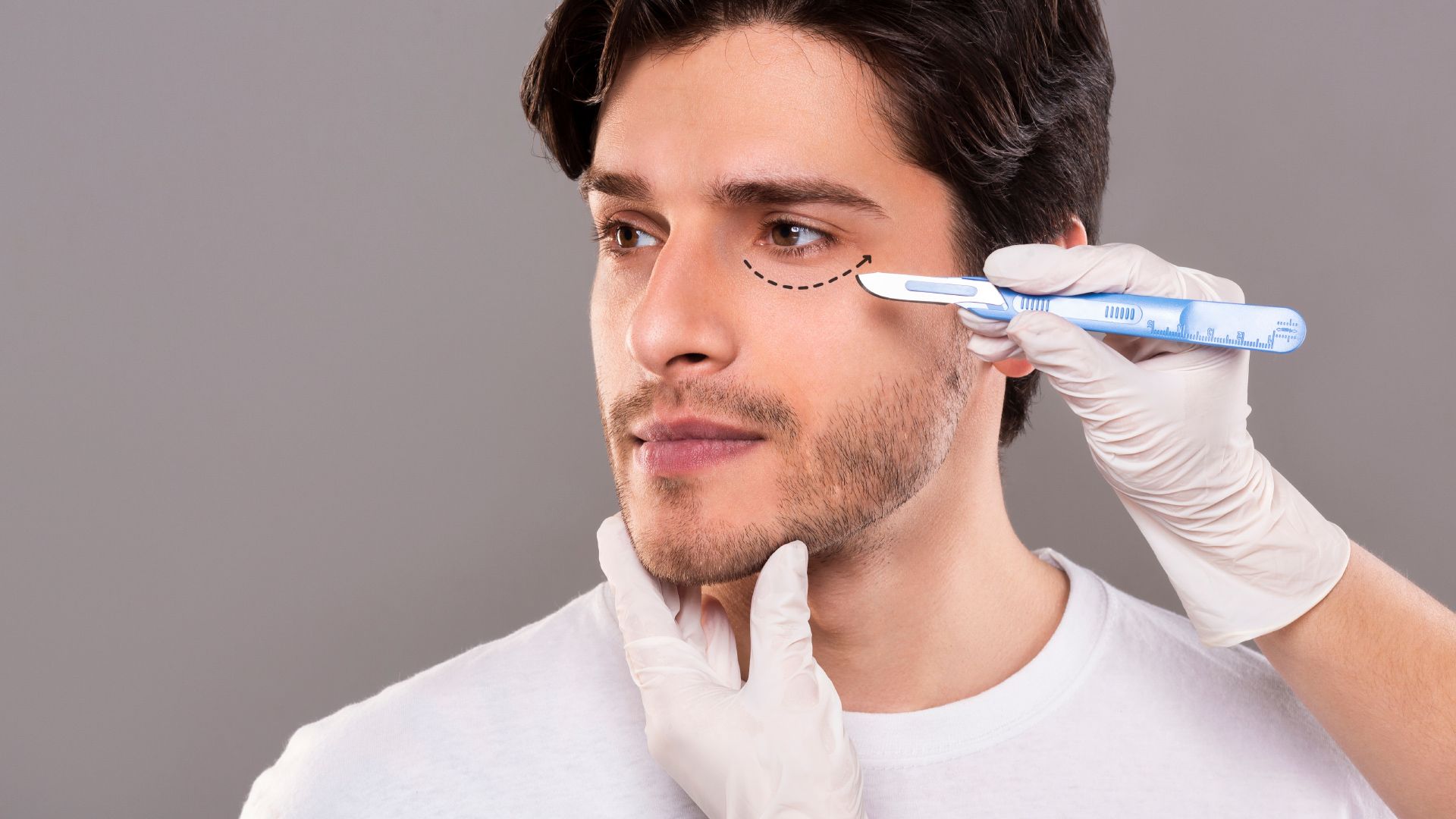
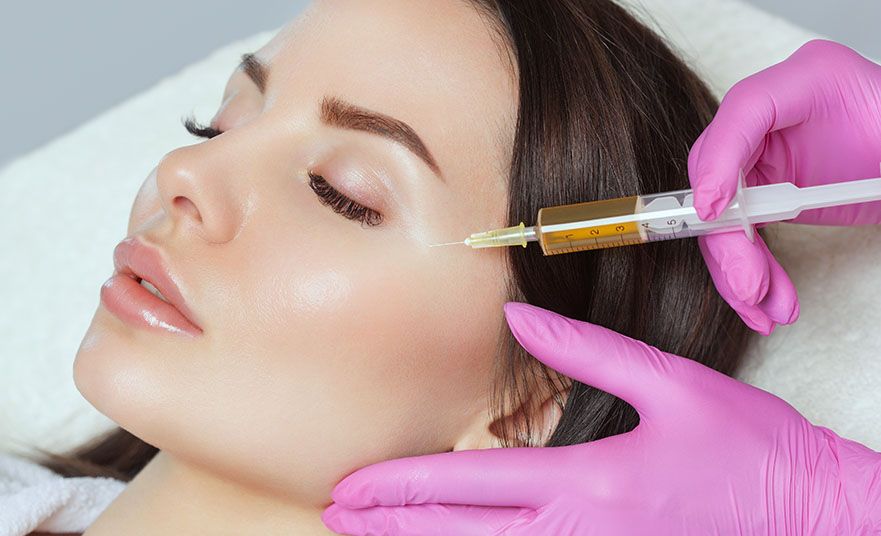
LET YOUR NATURAL BEAUTY SHINE WITH OUR COSMETIC EXPERTISE
Our cosmetic skin center offers a wide range of services, from botox, anti-aging treatments to acne solutions, all designed to help you feel confident and beauitful in your own skin.
Patient Testimonials
Over 300 5-Star Reviews


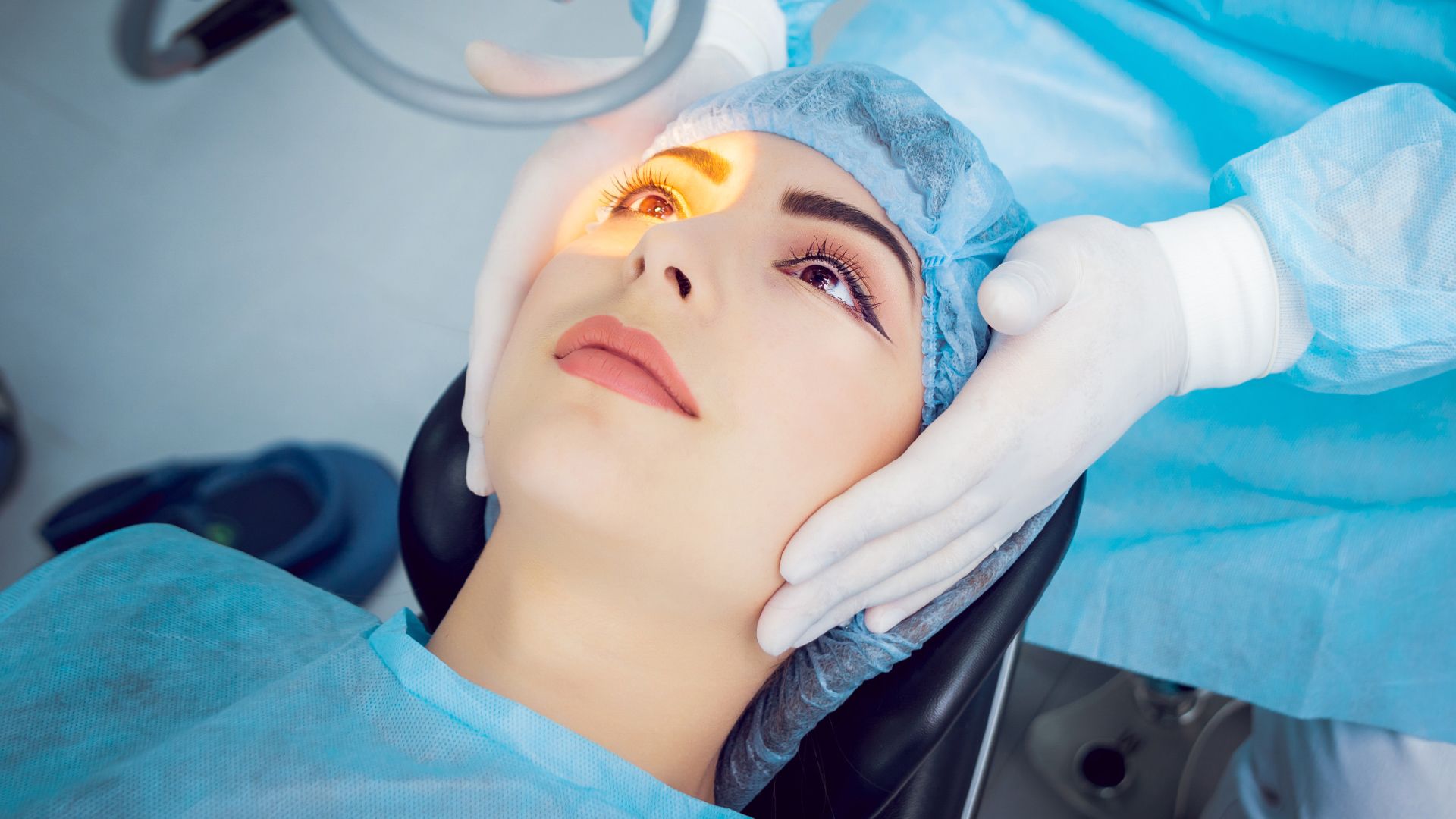
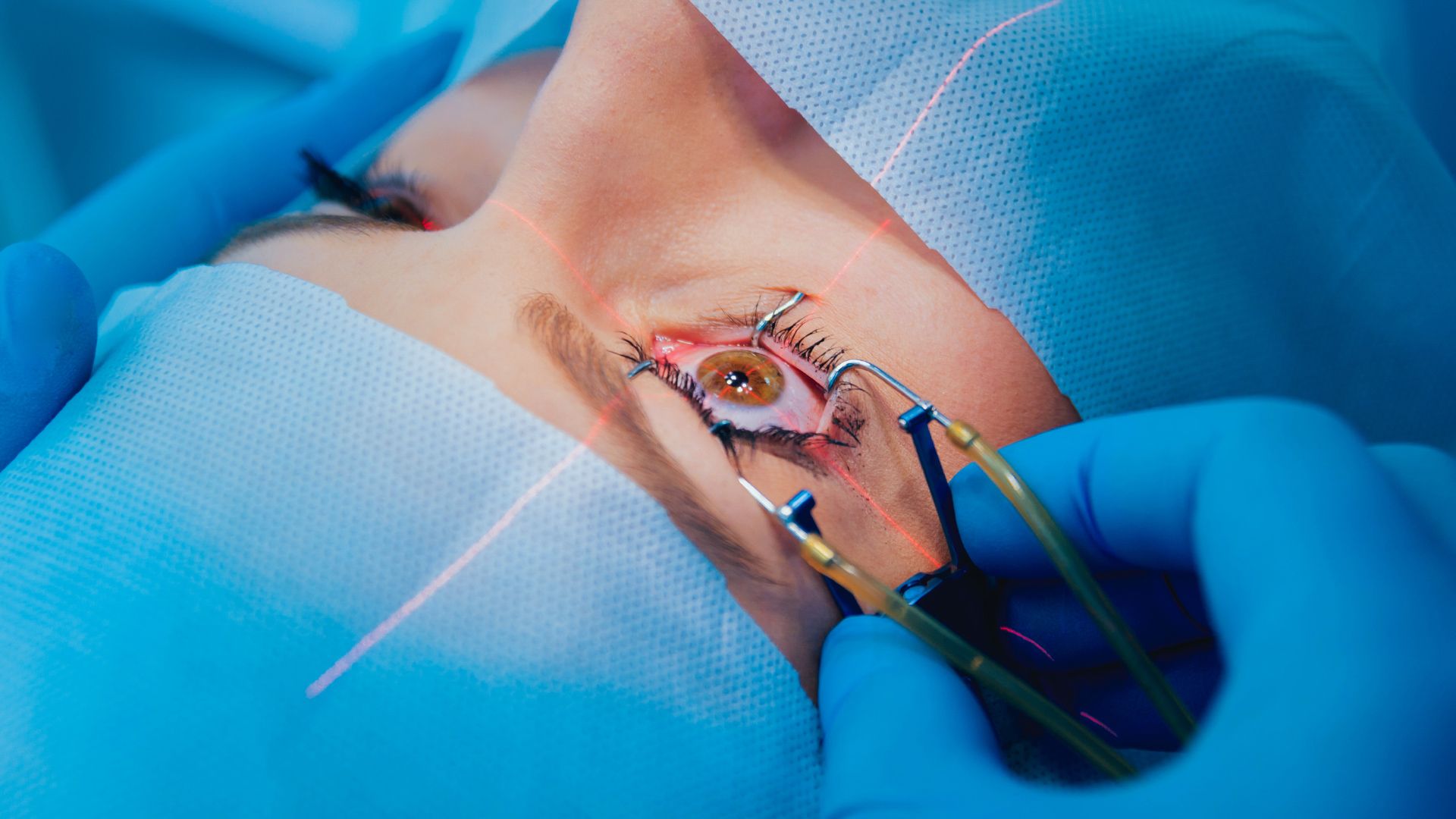







Both my husband and I are patients with Dr. McCabe. We absolutely love him and his staff. There have been some times where we have waited longer than usual but the visit and the work he does are always worth it. Dr. McCabe spends the time to talk with you, make you feel comfortable and to be sure you don't have any further questions. He has done an excellent with my left eye and two days after surgery I am feeling no pain and ready for the right eye. I wouldn't go anywhere else.
Jeanne K.
View All Reviews